
In a study published in Nature Communications on June 18, scientists from the Institute of Biophysics of the Chinese Academy of Sciences (CAS), Westlake University, and the Institute of Botany of CAS, used a combination of biochemical, mass spectrometry analysis, and cryo-electron microscopy techniques to discover a protein complex bound to the monomeric core complex of photosystem II (PSII) produced under high light conditions and named it the PSII-TPP complex.
PSII is the energy converter in the oxygenic photosynthetic system. The functional groups in PSII are highly susceptible to damage under light induction and by the action of reactive oxygen species generated during photosynthesis, leading to loss of activity. Damaged PSII must be restored to function through a complex biological process known as the PSII repair cycle, the molecular mechanisms of which are elusive and remain to be thorpughly elucidated.
In this study, the researchers identified the PSII-TPP complex as consisting of Thylakoid Enriched Factor 14 (TEF14), Photosystem II Repair Factor 1 (PRF1), Photosystem II Repair Factor 2 (PRF2), and an α-tocopherol quinone (α-TQ) molecule.
TEF14 is a soluble protein that binds to the luminal domain surface of the CP47 subunit, a core antenna complex of the PSII core. Studies suggest that TEF14 may be involved in the dissociation process of PsbO during the disassembly of the PSII supercomplex in the repair process.
PRF1 is a single-pass transmembrane protein, and the study suggests that this protein may be involved in promoting the dissociation of a peripheral antenna complex called CP29 from PSII during the PSII repair process. PRF2 is another single-pass transmembrane protein located near cytochrome b559, and its binding causes the plastoquinone diffusion channel in PSII to switch from an open to closed state.
α-Tocopherol quinone (α-TQ) is a small molecule compound that plays a critical role in plant photoprotection processes. For the first time, the researchers discovered that the α-TQ binding site near cytochrome b559, suggesting that α-TQ may prevent the transfer of electrons from cytochrome b559 to surrounding oxygen molecules through competitive oxidation, thereby preventing the generation of reactive oxygen species and protecting PSII.
Based on biochemical and structural analysis results, this study proposes a model for the functional mechanisms of TEF14, PRF1, PRF2, and α-TQ in the PSII repair cycle. This study has solved a puzzle in the molecular mechanisms of the PSII repair cycle, and it provides new insights into the disassembly and photoprotection processes during the repair of PSII complexes.
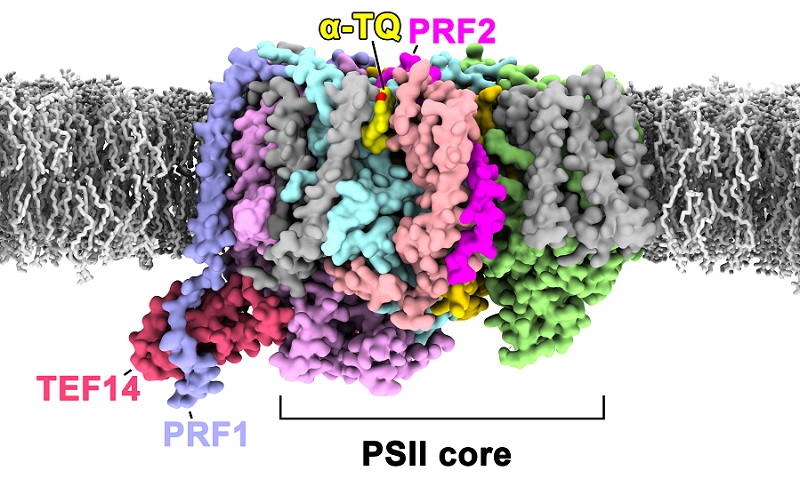
PSII-TPP complex formed during the early stages of PSII repair. (Image by LIU Zhenfeng's group)
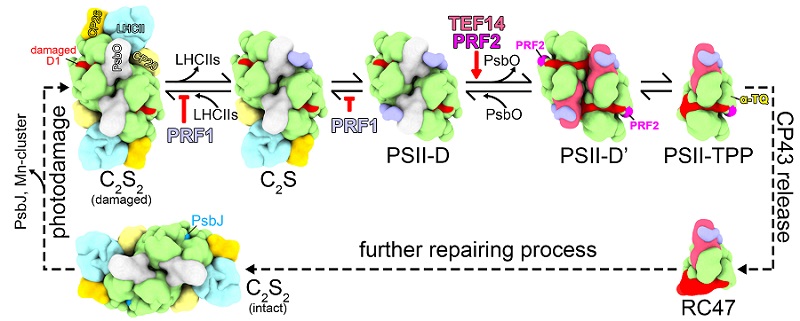
Model of the functional mechanisms of TEF14, PRF1, PRF2, and α-TQ in the early stages of PSII repair cycle. (Image by LIU Zhenfeng's group)

86-10-68597521 (day)
86-10-68597289 (night)

52 Sanlihe Rd., Xicheng District,
Beijing, China (100864)

